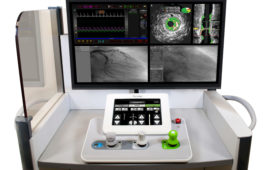
Corindus Vascular Robotics, whose CorPath GRX has FDA approval for percutaneous coronary and vascular procedures, will be acquired by Siemens Healthineers.
Corindus submits CorPath GRX to FDA for robotic neurovascular interventions
Corindus Vascular Robotics this week said it applied to the U.S. Food and Drug Administration for premarket clearance to use its CorPath GRX robotic surgical platform in neurovascular interventions. The Waltham, Mass.-based company has already received FDA clearance for percutaneous coronary interventions (PCI), which it won in 2016, and for peripheral vascular interventions (PVI), which it won last…