The obvious superiority of solid-state devices has rendered vacuum tubes obsolete in most applications. But an important exception is the photomultiplier tube. It is an elegant assembly of metal substructures in a glass envelope, providing unprecedented low noise and high amplification at generous bandwidth. The inexpensive device can actually detect a single photon that enters the envelope.
The photomultiplier’s working principles, Einstein’s photoelectric effect and secondary emission, are simple concepts, successfully combined by the Radio Corp. of America long before it was absorbed by General Electric and subsequently deconstructed. The 931 photomultiplier tube performed many functions — in astronomy, WWII radar jamming, medicine, and theoretical probes into the mysterious world of quantum mechanics. Its inner workings are straightforward:
The photoelectric effect, also called photoemission, arises when a body, struck by light, emits electrons. Moreover, alterations in the intensity or wavelength can result in changes in the rate of emission of the electron stream. Strangely, electrons are only emitted when the incident light exceeds a minimum frequency, regardless of the intensity or duration of the light. Albert Einstein explained this odd phenomenon by theorizing that the beam of light really consists of discrete quantized packets of photons. This, in conjunction with groundwork previously laid by Max Planck, was the intellectual event that gave rise to quantum mechanics. Planck had proposed what came to be called the Planck Relation, E = hv, which expressed energy E as a function of frequency v with a constant h factored in.
This is one of the underlying principles of the photomultiplier tube, the other being secondary emission: When electrons of sufficient energy strike a surface or pass through a conductive body, secondary particles are emitted into the surrounding space.
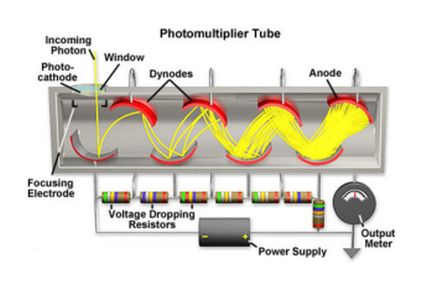
The National High Magnetic Field Laboratory gives this schematic for photomultiplier tube operation.
In a photomultiplier tube, electrons are emitted from a photocathode and accelerated in a beam that strikes the dynode, which is a polished metal electrode suitably biased. When this happens, through the miracle of secondary emission, a greater number of electrons, perhaps in the ratio of five to one, get released. They strike a second dynode, where again the number of emitted electrons is multiplied by the design factor.
By stacking a chosen number of dynodes in a photomultiplier tube and connecting the output to an auto-ranging oscilloscope, it is possible to see a measurable signal based on a single photon at the input.To observe the effect, it is necessary to have a device that will emit a single photon or a stream of photons separated by a significant time interval, or to access such a naturally occurring signal.

A real photomultiplier tube.
A device that can emit a single photon was shown to be problematic if not theoretically impossible by the Heisenberg Uncertainty Principle. That is because a single photon in theory cannot exist in nature. In fact, to prove or disprove Heisenberg’s basic thesis, a single photon source in conjunction with a device that can detect it (the photomultiplier tube) would be highly desirable. Accordingly, researchers sought a workaround that would allow them to emit, detect and measure single photons.
It is a relatively simple task to generate a signal that consists of packets containing an average of one photon or pairs of photons, but constructing a reliable source of single-photon emission is difficult although not impossible. One way it can be done is to generate two photons whose corresponding wavelengths are at slightly different frequencies, then minimize the bandwidth, in effect filtering out one of the photons. In this procedure, the second photon is not eliminated but just separated or sent off in a different direction.
Another major difficulty lies in the fact that the signal is incredibly weak. A typical low-level signal seen in the world of electromagnetic radiation may consist of 1023 photons landing each second, so this should put the single-photon quest in perspective.
Recently a team of scientists at the University of Basel in Switzerland has shown that the obfuscation of photons is reduced by the indeterminate spin of quantum dot atoms. This means that it is possible to control the precise frequency corresponding to otherwise fluctuating photon emission.
A quantum dot is a unit of several hundred thousand atoms that is capable of exhibiting qualities of a semiconductor material. That being the case, creation of a reliable and consistent single-photon source is envisioned. Because the diameter of a quantum dot is in the low nanometer range, its free charge carriers experience quantum confinement in every spatial dimension. Band gap and emission frequency become highly tunable, allowing for single-photon differentiation, which is precisely what is needed to achieve one-photon emission.
Further developments in this area will lead to a greater understanding of the quantum realm that is at once everywhere and yet highly elusive.
The post The basics of photomultiplier tubes appeared first on Test & Measurement Tips.
Filed Under: Test & Measurement Tips