Swiss-based icotec AG developed industrial applications for a composite flow molding method using a fiber composite made of high-tech plastics that is strengthened with more than 50% carbon fibers. Highly resilient products can be made from it. They are corrosive-free, can be electrical and thermal isolating, and do not cause signal errors. These properties are important for the development of medical implants. In fact, the company is working on a new spine implant to replace damaged spinal discs.
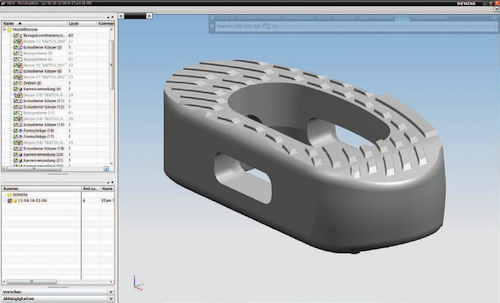
Designers at icotec use NX software to generate complex, free-form part surfaces. The company serves the civil and military aviation and life science markets.
After creating the first designs using NX software and preparing static-mechanical calculations using STL file format data, icotec delivered prototypes for evaluation by a surgical team. Concurrently, surgical operating tools were designed. According to Ramon Huppi, project manager, “Due to the NX’s synchronous technology, we have more possibilities to move surfaces and define manufacturing ready parts.”
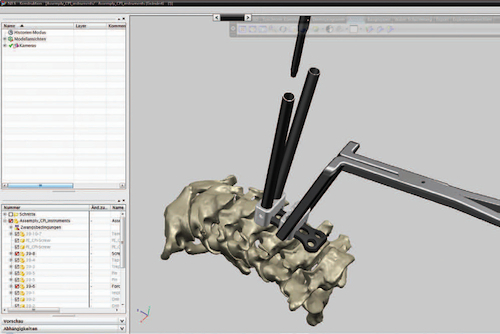
NX users rely on its synchronous technology and full-screen capabilities to design a next-generation spinal implant.
For example, different sizes and modifications for the cervical spine quickly resulted in a large product family. The tool design team compensated for this variety using an efficient inlay technique. Huppi added, “We sent a Parasolid file to our service provider who used it as the starting point for the programming. Once the first samples were made using one of icotec’s two continuous flow manufacturing (CFM) machines, the approval tests were conducted. Static and dynamic tests as well as studies under compression, torsion, and shear were performed by external test laboratories in up to five million cycles. Changes were done quickly using synchronous technology.”

icotec uses an innovative new carbon-based composite material to create rotor fastening screws for helicopters, retaining brackets for airplane luggage racks, and biomedical parts.
New products for aviation and life sciences, such as the implant, must comply with the strictest development and approval standards. Comprehensive requirements pertaining to quality testing, documentation, and traceability for every part must be fulfilled. To address compliance, icotec uses Teamcenter software for product data management. The software handles all aspects of part management. This includes controlled check-in and check-out directly in the CAD system and searching for precursors, revision status, and similar projects. It also handles a comprehensive and daily drawing management process for the development team. In the future, the company will use Teamcenter to support the approval processes for the American Food and Drug Administration. It also plans to start leveraging Teamcenter’s data organization function to streamline the management of more than 30 years of aircraft spare part inventories and warranty obligations.
Siemens
www.siemens.com/plm
Filed Under: Data acquisition + DAQ modules, ENGINEERING SOFTWARE





Tell Us What You Think!