The chemical battery resembles a capacitor in the sense that both are capable of taking a charge, storing electrical energy and releasing it later as needed, simply by connection to a load. But these devices work entirely differently. A capacitor can be idealized as consisting of two conductive plates connected to wire leads, separated by a dielectric material such as paper or mica. Capacitance, a measure of the ability to store a charge, depends on the area of plates, their proximity, and the dielectric constant of the material that separates them. The electric charge is actually stored in the dielectric material.
A chemical battery also stores a charge, but the mechanism is entirely different. It is an assembly of wet (liquid electrolyte) or dry cells (paste electrolyte) that is intended to accept, hold and release as needed, an electric charge. As in capacitors, parallel plates can serve as electrodes. The material between them holds the charge and is known as the electrolyte. The electrolyte changes form as the battery charges or discharges. (Secondary batteries are rechargeable. Primary batteries are not; their chemical reactions don’t reverse easily and their active materials can’t return to their original states.) For example, in the familiar 12-V lead-acid battery used in automotive applications, the pH level changes as the battery charges or discharges. When fully discharged, the electrolyte is close to water. When fully charged, it is a strong acid.

The lead-acid battery was invented in 1859 by French physicist Gaston Planté. It is the oldest but not the only type of rechargeable battery. Others include:
Nickel-metal hydride – NiMH has replaced the highly toxic nickel-cadmium (NiCd) battery. Also, the NiMH battery has higher capacity. Batteries and chargers of this type are common and readily available. They are suitable for high-drain applications, where lots of power is needed quickly, as in digital cameras. A disadvantage is the low non-standard voltage (1.2 V). This is not always a problem, but unregulated loads such as light bulbs will run dim. NiMH batteries retain full voltage throughout most of the charge cycle, then drop off abruptly.
Nickel-zinc – NiZn batteries are good candidates for high-drain applications, lasting longer than NiMH units. The nonstandard voltage (over 1.65 V) means that flashlight bulbs burn slightly brighter except for LED flashlights, which are regulated. These batteries self-discharge rapidly, making for short shelf life. They also need a specialized charger and are available only in AA and AAA sizes. There have been reliability problems with NiZn cells. With these drawbacks they are to be avoided for most uses.
Lthium Ion (Li-ion) batteries can be deceptive because the voltages are nonstandard, except in the 9-V unit. AAA, AA, C and D cells all output 3.7 V rather than the standard 1.5 V. So if they are inadvertently inserted into standard battery-powered devices, they may damage the device. Moreover, except for the 9-V Li-ion, special chargers are needed. That being said, in the correct application these batteries have some superb qualities. They work well for high-drain loads such as digital cameras with flash and laptop computers. Li-ion batteries are also used extensively in ever-higher-voltage cordless tools.
Alkaline batteries are available in rechargeable and non-rechargeable versions. They are noted for a slow self-discharge rate, which translates into long shelf life. Unfortunately there are some downsides. One is that the battery capacity drops following each cycle. They are not suitable in high-drain devices such as digital cameras and laptops. Also there is significant risk of leakage and corrosion damage. Because of low initial cost (compared to other rechargeables) they are suitable for inexpensive devices such as flashlights where corrosion damage will not be a life-changing event.
Finally, a few words about a special case of energy storage known as the Baghdad battery. It was thought that the electrochemical battery was unknown prior to its invention in 1800 by Alessandro Volta. This belief was called into question when Wilhelm König discovered some curious artifacts in the National Museum of Iraq where he worked as a painter in the 1930s. Returning to Germany in 1940 due to sickness, he wrote a paper describing his find and theorizing that they might have been a galvanic cell.
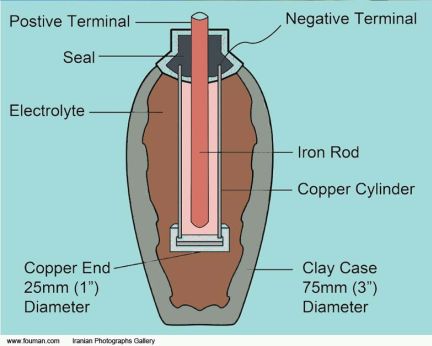
The objects consisted of a ceramic pot, about five inches high, a copper cylinder and iron rod, both of which could fit inside the vessel, held in place and separated from each other by a plug made of bitumen, which happens to be an electrical insulator.
König believed the artifacts originated in the Parthian Period, ranging from 250 BC to 224 AD. Unfortunately, the time and circumstances of the vessel’s original discovery, prior to its placement in the National Museum of Iraq, are uncertain, and precise dating is difficult. The style of the pottery would seem to indicate that it was formed during the Sassanid Period, 224 to 640 AD.
König believed these objects constituted a single galvanic cell. A liquid acidic electrolyte, of course, would have to be poured into the vessel. Vinegar, wine, grape juice, lemon juice and other fairly weak but effective acids were available. Highly conductive metals such as copper, gold and silver could readily be drawn to form wires so multiple cells could be connected in series and parallel configurations, and the power conveyed to a load. However, there is no evidence that electrical wire was even conceived at the time.
Over the years, critics have gone to great lengths to challenge König’s hypothesis that the vessel was intended as a galvanic cell. Did the ancients use electrical power? In view of the construction, it seems obvious that was its purpose. It has been proposed that the hookup was used to electroplate metal objects. A thin, uniform layer of gold could be applied, and electroplating would have been more efficient than other methods that were used at the time, such as application by hand using a mercury paste.
Critics have pointed out that the use of the Baghdad vessel to generate electrical power by chemical means would presuppose a high degree of technical knowledge. Is this difficult to envision, in view of the great capabilities of our ancestors, demonstrated by their abilities to construct huge pyramids and great buildings?
The post Basics of chemical batteries appeared first on Test & Measurement Tips.
![]()
Filed Under: Capacitors, Test & Measurement Tips