By Keith Halbert, PhD • Portescap || Device makers and surgeons have for decades relied on sterilizable brushless direct current (BLDC) motors to meet the torque, speed, and reliability requirements of surgical handtools. Now surgical robotics are leveraging these same capabilities.
Today’s surgical robotics and robotically-assisted surgical devices often require BLDC motors to meet demanding requirements. While motors and motion are core to all robotics, surgical robotics demand the capabilities of both traditional robotics and traditional surgical handtools. In addition, devices used in surgery must be sterile. Often these devices must reliably and consistently function despite repeated steam sterilization during reprocessing, in addition to demanding field use.
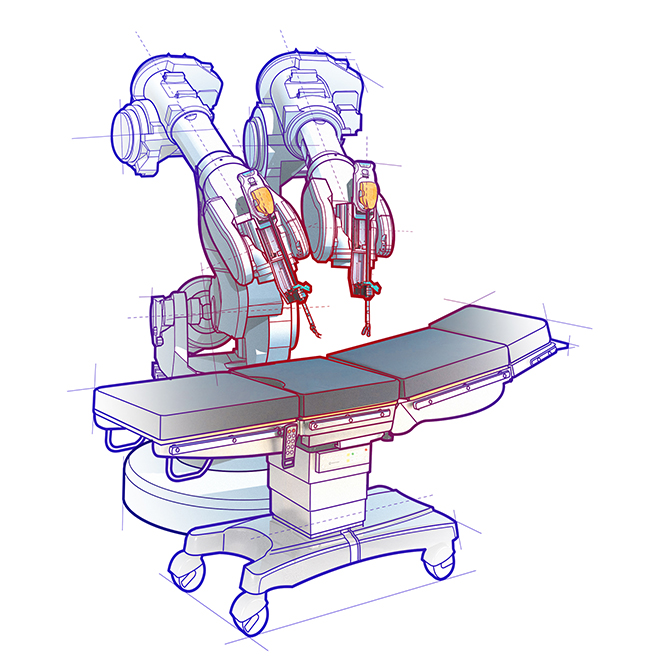
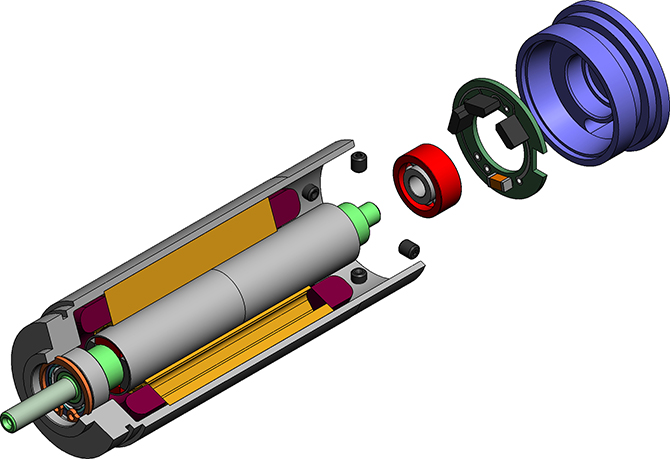
Shown here is an example of a surgical robot. Portescap tailors motors to such designs with customized shaft cannulation, motor electromagnetics, mounting features, gear ratios, and pin connections (instead of flying leads). In short, Portescap’s expert design engineers collaborate with device engineers to customize these and other features to the unique requirements surgical handtools or surgical robotics.
Beyond reliability requirements, surgical device designers must also ensure their end products satisfy exact speed and torque requirements, ability to withstand the high temperatures of sterilization, remain cool during operation, and meet extreme positioning demands. These device makers need motion components that are suitable for their application, and which have been appropriately customized to both integrate with their tool and make the proper tradeoffs to optimize performance. Methods to maintain and preserve the sterile field in the operating room to prevent infection, cross contamination, and the spread of disease are all critical concerns.
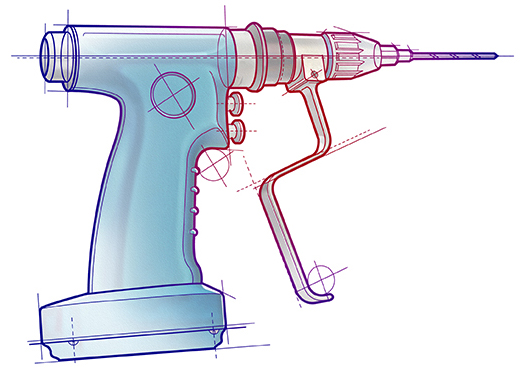
Shown here is an example of a powered surgical handtool so essential in operating rooms. Portescap has supplied highly specialized motors for these designs as well as arthroscopic shavers, sagittal and oscillating saws, medium and high-speed drills, other orthopedic drills, wire drivers, and surgical staplers.
Here are the most common approaches to achieve the required sterilization:
The disposable tool: In some cases, hospitals and surgeons elect to use disposable, single-use tools. These generally employ inexpensive motors (given long life isn’t required) and often plastic components. Such tools must be discarded after each surgery.
While this approach simplifies reprocessing and eliminates the requirements of tool maintenance, it also necessitates a consistent supply of tools be maintained and increases the amount of hazardous waste produced by the hospital. In addition, disposable tools aren’t typically the most economical option when considering the total cost to the hospital.
Modular design for sterilization using non-sterilizable components: Another approach is to design devices so that exposed components are sterilized and others are not. Here, a design may house the motor, controller, and battery pack inside an enclosure — and hospital staff remove the motor and battery pack from the tool prior to sterilization. This approach requires that a special process is correctly followed to ensure the reprocessed tool is properly sterilized and may also require more durable electronic components and connections in the design due to repeated disconnection and reconnection of the motor and battery from the system.
Protective barriers: Yet another approach is to cover the robotic arm or instrument with a (typically disposable) sterile barrier — for example, a plastic draping or plastic clamshell. When successfully executed, such barriers maintain a sterile field and eliminate the need for components outside the field to be reprocessed. This approach is a common with large surgical robotic systems — for which autoclaving of the entire system is impractical.
The ergonomic requirements of robotic systems also differ from those of traditional handtools. More specifically, the motor may be physically located away from the surgical end effector and transmit motion via cable-drive — which may not be feasible for traditional surgery when a surgeon must precisely manipulate a handtool to perform a delicate task.
This design approach is also common for medical procedures having less stringent sterilization requirements — such as dental and tattoo applications, for example. But a downside includes complex draping schemes needing systematic removal and replacement, which can significantly increase the time the operating room is engaged for a surgery. Draping can also be bulky and awkward and can reduce visibility in the surgical theater — in turn degrading the effectiveness of the surgeon.
Autoclavable motors: Medical devices can also be designed utilizing only sterilizable components. In such designs, the motors must be sterilizable. The introduction of sterilizable BLDC motors three decades ago let tool designers produce high-power ergonomic tools that could be trusted to be sterile due to the entire tool having gone through the sterilization process. The benefits carry over to robotically assisted surgical devices, most of which also necessitate sterile designs of small size, high power, durability, efficiency, low noise, and long life.
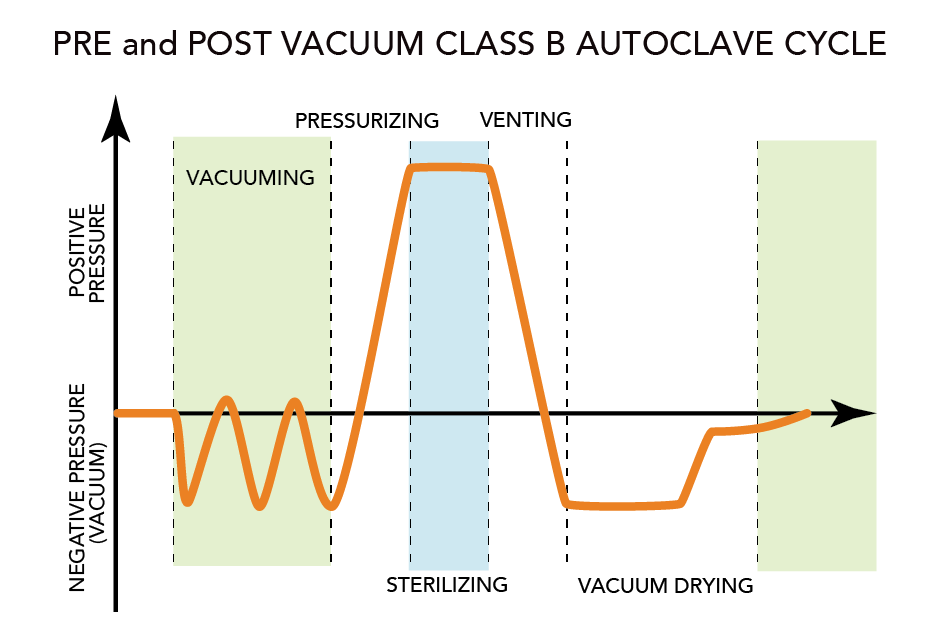
What is an autoclave cycle?
The most common sterilization method used in hospitals is autoclaving, also called steam sterilization. During autoclaving, surgical handtools are exposed to 100% humidity, 135ºC (275ºF) and pressure variations for up to 18 minutes. Most autoclaves also have additional vacuum cycles to facilitate steam penetration and kill viruses, fungi, bacteria, and spores that can hide in microscopic cavities on the device. Repeated exposure to this environment is what typically causes significant electrical and corrosion problems for motors and devices insufficiently designed to withstand these conditions.
Autoclavable BLDC motor design for surgical devices
Both traditional motorized handtools and robotically assisted surgical devices can use BLDC technology in either a slotted or slotless configuration. Note that slotted versus slotless refers to the lamination type in the stator of the motor. Both technologies have their strengths, and the application requirements dictate which technology is better suited to the design at hand.
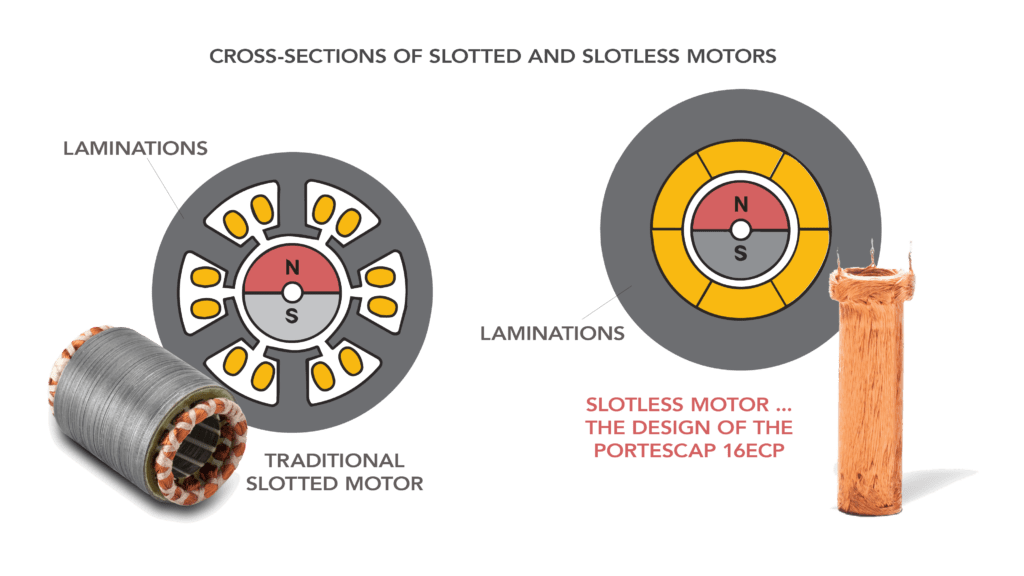
Shown here are cross-sections of slotted and slotless motors for precise motion control. Traditional slotted motors have the space for winding protection in the form of coating molding. Slotless motors offer other benefits but allow no such room for winding protection.
Slotted BLDC technology has been a proven solution in the surgical motor market for more than 30 years. In slotted motors, the copper coils are wound within the slots of the lamination stack — so are inherently protected. Additional insulation layers and molding material can easily be added without impacting motor performance. This physical configuration makes slotted BLDC excel in miniature motion designs rugged enough to withstand harsh environmental conditions, such as those seen in autoclave or during surgeries that expose the motor to saline and other contaminants.
In addition, the slotted design provides:
- Easy customization to electromagnetics (windings, lamination stack length , and so on)
• Very high dielectric resistance (1,600 V AC hi-pot or higher)
• Improved heat dissipation and thus higher continuous torque
• A small magnetic air gap to allow use of thinner magnets and a higher permanence coefficient (which imparts torque stability over a large temperature range)
• Lower rotor inertia than slotless BLDC motors
Slotless motors are the other BLDC motor technology. These motors are also very capable and may be well suited to the application. In slotless motors, the coil is wound in a separate external operation and is a self-supporting subcomponent.

This is a brushless dc slotless motor.
This rigid coil is then inserted directly into the air gap during motor assembly. In this design, the magnetic induction in the coil is decreased since the air gap is large. Induction in such a motor is usually much smaller than in a slotted BLDC motor, so a larger and more powerful magnet is typically needed to compensate for the loss of induction.
While slotless motors can be designed to withstand steam sterilization through insulation and other protective coatings on the exposed electronic components, achieving long-lasting and dependable protection from harsh environmental conditions is inherently more challenging when compared to a slotted motor. If autoclavability or very high numbers of sterilization cycles are not needed — there are aspects to a slotless design that may be an advantage for a given application: zero detent torque (no cogging), smooth operation at very high speeds, increased motor inertia, and high peak torque capability.
This is a Portescap Size 9 brushless dc slotted motor.
Some surgical procedures or device applications demand high-precision control of the motor. This is true of robotically assisted surgical devices using sophisticated sensors, vision systems, haptic feedback, or 3D mapping to target material manipulation at the sub-millimeter level. Successful execution of the surgery may need extremely high precision control of the motor output. The precision requirements may go beyond that which is delivered by traditional Hall sensors, which can detect rotor position in 60° increments. Using an encoder can provide feedback for control of speed and positioning of the rotor at << 1° increments.
Encoders provide angular position measurements of the rotor shaft at a much higher precision than three Hall sensors can provide. Such feedback supports position control and better BLDC-motor-control accuracy than otherwise possible. From the position measurements the encoders measure, both acceleration and direction can be inferred.
Tip: When specifying an encoder, first determine required accuracy and resolution. The technology type must also be chosen. Optical and magnetic are the most common technologies in rotary encoders.
In autoclavable applications such as surgical tools, magnetic encoders are typically a robust and reliable option. Incremental or absolute feedback are two common variations for communicating the angle value. If using incremental signals an index pulse, once per revolution, and a counter is needed to calculate the absolute angular position. Otherwise the feedback is relative.

This is an autoclavable encoder.
Absolute feedback is typically serial communication such as SSI, SPI, or BiSS to provide an encoded angle value between 0° and 360°. Options from Portescap include:
• Sterilizable option — designed and tested to more than 2,000 autoclave cycles
• Hall sensor signals for 6-Step commutation (U, V, W)
• 10-Bit incremental encoder (A, B, Z)
• 11-Bit resolution absolute angle encoder
• Absolute position output via SPI
• Differential output for noisy environments
• Off-axis mounting allowing for cannulation
Modern surgical devices — both traditional handtools and robotically-assisted devices — have extremely demanding and exact motion requirements. Those requirements can be met by working with a motor supplier with the necessary technologies and experience with both traditional surgical handtools and robotically assisted surgical devices.
Portescap | www.portescap.com
Filed Under: Fighting COVID-19, Motion Control Tips




